And anionic #Cl^-# should be larger than #Na^+#, with respect to their parent atoms. 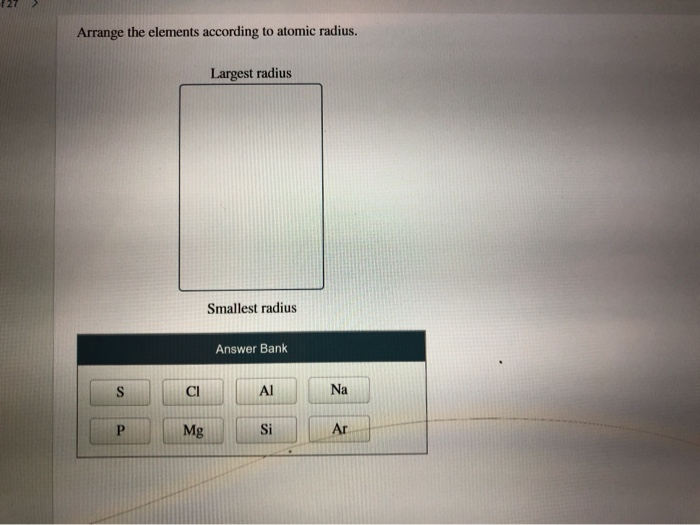
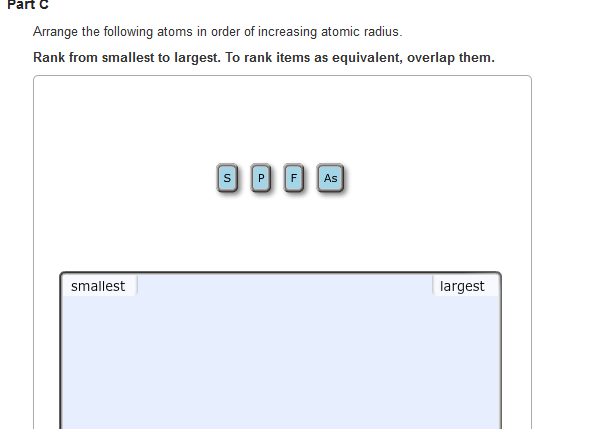
Atomic #Cl# should be smaller than atomic #Na#, given their position on Period 3. Does the diagram support this conjecture? Why or why not?Īnd in fact many Periodic properties may be rationalized on this contest between nucular charge, and shielding by other electrons.Īnd so to your question, finally! #Na^+# is smaller than #Na# and #Cl^-# is larger than #Cl#. And since they accept electrons, the ionic radius of the ANION, should be LARGER than the parent atom. On the other, other hand, non-metals are oxidizing, that is they accept electrons to form negatively charged ions. Metals are electron rich and REDUCING, and form positively charged cations and thus, since the valence electrons are removed, their ions should be of reduced radius. On the other hand ionic radius depends on the nature of the element: metal or non-metal. Once the shell is full, however, shielding of nuclear charge becomes relatively effective, and the next Period builds on a new valence shell that is farther removed from the nucleus. arrange each set of atoms in order of increasing radius: (b) S, Si, Sr. And thus across the Period atomic size decreases. Well, atomic size depends on electronic radius, and INCOMPLETE electronic shells shield the nuclear charge very ineffectively. Largest radius Smallest radius Answer Bank Be Mg Mg Rb Ca Arrange the atom and ions according to radius. Largest Smallest Answer Bank oxygen silicon calcium barium Arrange these elements according to atomic radius. OK OLA Determine whether Se or Ga that has the more negative (more exothermic) electron affinity. Largest radius Smallest radius Answer Bank Ar Si Mg Al Na Arrange the elements according to atomic radius, from largest to smallest. 
Largest radius Smallest radius Answer Bank As Ge Kr Ca K Br Se Ga Determine whether Li or K that has the more negative (more exothermic) electron affinity. We know that (i) atomic radius decreases ACROSS a Period from left to right as we face the Table and (ii) atomic radius INCREASES down a Group, a column of the Periodic Table. Transcribed image text: Arrange the elements according to atomic radius. a group the elements become more reactive this is because the atomic radius increases the. Construct Mohr's circle for an element in biaxial stress subjected to two equal stresses x y 0 as shown in the figure on page 449. (b) Elements of group 17, in decreasing order of reactivity. And the only reasonable definition is by ATOMIC or IONIC RADIUS. Construct Mohr&39s circle for an element in biaxial stress subjected to two equal stresses. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
